Folate, colloquially known as Vitamin B9, stands as an indispensable nutrient and a foundational pillar of human biology. Its roles are elemental, facilitating DNA synthesis and regulating cellular repair. It also manages the detoxification of harmful metabolic byproducts. Yet, the common understanding of this essential vitamin is fraught with a critical misconception. This error pervades the medical literature. In public consciousness, synthetic folic acid is treated as synonymous with natural folate. This conflation ignores basic chemistry. This error is not merely semantic; it represents a profound biochemical misunderstanding with significant health consequences. For many, synthetic folic acid remains a metabolic challenge. It lingers unutilized in the bloodstream. Meanwhile, cells starve for the specific compound they truly require: activated folate.
This essay explores the critical distinction between folic acid and activated folate through the lens of modern biochemistry. The active form is scientifically known as 5-Methyltetrahydrofolate (5-MTHF). We examine the intricate metabolic pathways of the one-carbon cycle and the MTHFR genetic bottleneck. This builds a robust case for a new paradigm in nutritional science. It moves beyond the blunt instrument of mass fortification with folic acid. It favors a personalized, biochemically informed approach. The goal is providing the body with the bio-available Vitamin B9 it needs to thrive. The journey to a targeted nutritional strategy begins with a deep appreciation for this nutrient. This exploration illuminates why activated folate serves as the superior choice for optimal health. It bypasses the metabolic hurdles that render other forms of B9 ineffective for many.
Molecular Distinctions in the Vitamin B9 Family
Grasping Vitamin B9 superiority requires understanding the metabolic journey from ingestion to cellular function. Folate refers to a family of compounds sharing a structural backbone, rather than a single molecule.In nature, leafy green vegetables provide dietary folates as complex polyglutamate structures. Upon consumption, gut enzymes deconjugate these chains into simpler monoglutamates, which the bloodstream then absorbs. Once absorbed, the liver performs enzymatic conversions to transform natural folates into the final, usable form.This multi-step process creates potential inefficiency, as the body often struggles to produce sufficient activated folate. This failure to fuel biological systems adequately undermines health.
The introduction of synthetic folic acid presents a far more significant metabolic challenge because it differs chemically from what the body expects. Folic acid is a fully oxidized, man-made molecule that does not occur anywhere in nature; manufacturers synthesize it primarily for its shelf-stability. Officials selected it as an ideal candidate for fortifying processed foods like flour to prevent deficiency diseases. To become biologically useful, however, an enzyme called dihydrofolate reductase (DHFR) must first reduce the folic acid. Herein lies the critical bottleneck that most medical professionals overlook. In humans, the activity of the DHFR enzyme is remarkably low, functioning several hundred times slower than in species like rats. Consequently, when the body encounters high doses of folic acid, the liver’s limited capacity is quickly overwhelmed, significantly halting the necessary production of activated folate.
The Metabolic Superiority of Active Folate
This enzymatic traffic jam causes Unmetabolized Folic Acid (UMFA) to accumulate in the systemic circulation. Research suggests potential links between high UMFA levels, masked Vitamin B12 deficiency, and increased cancer risk.The presence of UMFA signifies a profound metabolic failure where the body possesses high amounts of the wrong molecule. The body receives a precursor it cannot process, while methyl-dependent cells and tissues go without. Cells require activated folate, not folic acid, to drive their functions. Without this specific form, cellular machinery grinds to a halt, causing fatigue, brain fog, and systemic inflammation.
Supplementing directly with active folate bypasses the compromised DHFR pathway. As the primary blood plasma folate, it crosses the blood-brain barrier and enters cells directly without further reduction.This biologically efficient form makes its superiority over folic acid a matter of fundamental biochemistry. Without sufficient activated folate, the body compromises critical processes like neurotransmitter synthesis and DNA methylation. Therefore, individuals must ensure an adequate intake of this specific nutrient. The diagram above illustrates the metabolic blockage from folic acid compared to the direct 5-MTHF utilization pathway. This demonstrates why the active form maximizes cellular metabolism efficiency.
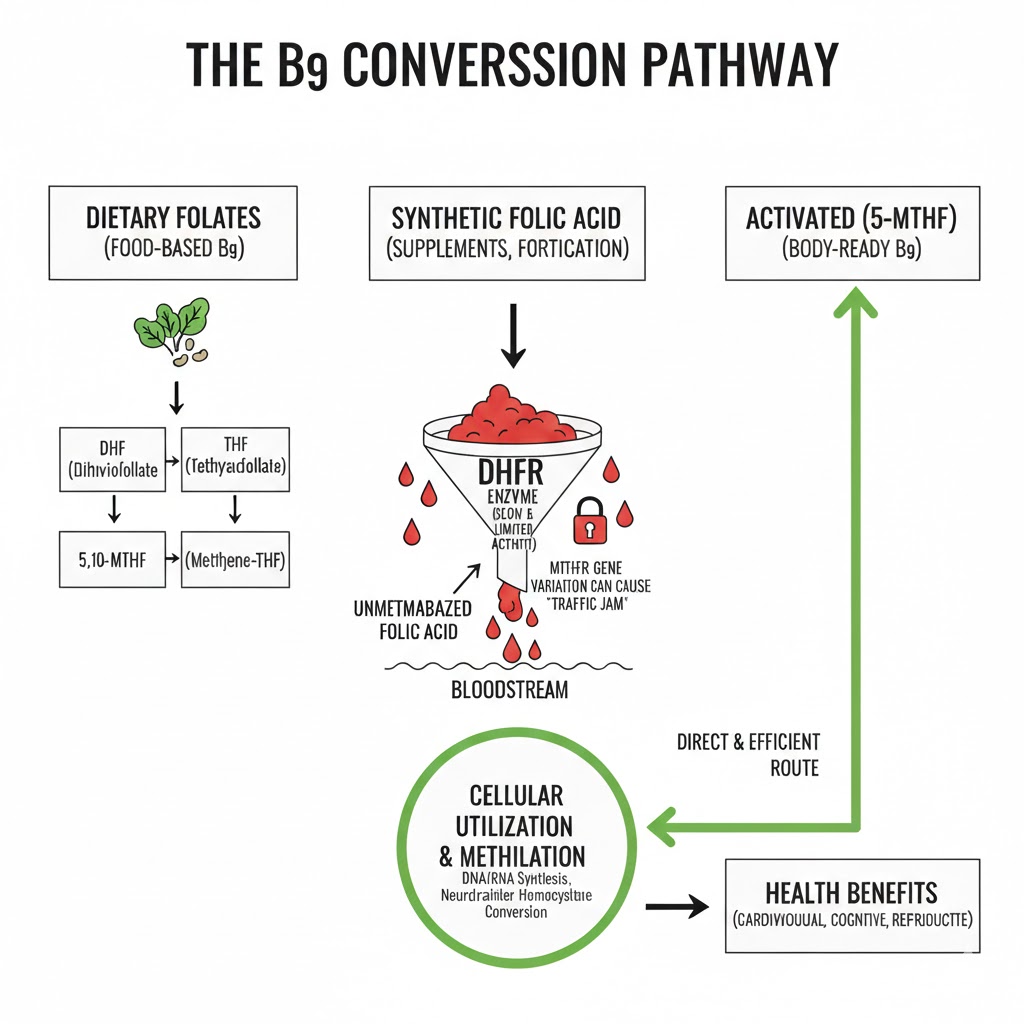
The Genetic Bottleneck of the MTHFR Mutation
Modern genomics and DNA sequencing powerfully reinforce the biochemical argument for prioritizing activated folate. Widespread genetic testing reveals that a significant percentage of the global population possesses common MTHFR gene variants.This gene provides the blueprint for the methylenetetrahydrofolate reductase enzyme, which converts folate into active 5-MTHF. Genetic variance compromises this enzyme, dramatically reducing the body’s ability to produce activated folate regardless of B9 intake. Even individuals eating folate-rich diets may suffer functional deficiency if their genetics hinder this final conversion step. These individuals require an exogenous source of the active nutrient to bypass metabolic hurdles.
Two primary MTHFR SNPs, C677T and A1298C, cause clinical concern by altering the enzyme’s structure and thermal stability. The C677T variant influences cardiovascular health and homocysteine regulation, acting as a major risk factor for heart disease.Individuals who are homozygous for this variant (possessing two copies of the gene) experience a staggering drop in MTHFR enzyme efficiency, often by as much as 70% compared to the wild type. The A1298C variant, meanwhile, is more closely linked to the production of neurotransmitters and consequently impacts mood and cognitive function. A person can be heterozygous (one copy) or homozygous for either variant, or be a compound heterozygote (one copy of each), all leading to varying degrees of reduced enzyme function. In all these scenarios, the body struggles to manufacture enough activated folate to meet systemic demands, creating a chronic deficit.
Systemic Consequences of Low Active Folate
The downstream consequences of this genetic bottleneck are systemic, severe, and affect nearly every organ system in the human body. A dysfunctional MTHFR enzyme stalls the entire “methylation cycle,” also known as one-carbon metabolism, which is the engine for cellular renewal. Methylation is a deceptively simple yet profoundly important biochemical process involving the transfer of a methyl group (one carbon and three hydrogen atoms) from one molecule to another. This transfer acts as a biological switch, turning genes on and off (epigenetics), repairing DNA, processing hormones, and detoxifying the body. When the production of activated folate is impaired due to an MTHFR mutation, this fundamental switch effectively rusts in place. Without the donation of methyl groups, the body loses its ability to regulate essential functions, leaving the individual vulnerable to accelerated aging and disease progression caused by unchecked oxidative stress and cellular damage.
Without an adequate supply of methyl groups derived specifically from activated folate, the body cannot properly convert the inflammatory amino acid homocysteine back into methionine. It cannot effectively synthesize neurotransmitters, nor can it manage gene expression or efficiently clear environmental toxins. Chronic inflammation, cardiovascular disease, and neurological dysfunction often follow as the methylation cycle slows down. For individuals with these genetic variants, consuming folic acid is akin to pouring crude oil into a car that requires refined gasoline; the tank is full, but the car will not run. The raw material is present, but the engine lacks the machinery to process it. Supplementing directly with activated folate is the only logical and effective solution, providing the finished fuel that the body’s compromised metabolic engine desperately needs. This makes a strong case for prioritizing this nutrient in all modern supplementation strategies.
Neuropsychiatry and the Role of Activated Folate
The recognition of activated folate as the body’s preferred form of B9 has catalyzed a revolution in clinical practice, particularly regarding mental health. Researchers and practitioners now leverage its unique properties to address several high-stakes health conditions where traditional therapies have failed. The shift from prescribing folic acid to utilizing 5-MTHF has demonstrated profound efficacy, particularly in the fields of neuropsychiatry and mood regulation. In the context of mental health, the brain has an immense demand for methyl donors to synthesize BH4 (Tetrahydrobiopterin), a critical cofactor required for the production of key neurotransmitters, including serotonin, dopamine, and melatonin. Without sufficient BH4, neurotransmitter synthesis plummets, leading to mood destabilization. This biochemical dependency explains why deficiencies in activated folate are so frequently observed in psychiatric patients.
In treatment-resistant depression, a central nervous system deficiency of active folate often underlies the condition. Standard antidepressants keep existing serotonin in the synaptic cleft but cannot help the brain produce more.If the brain cannot manufacture serotonin due to poor methylation, antidepressants have nothing to work with. Clinicians can restore neurotransmitter building blocks by providing 5-MTHF, which readily crosses the blood-brain barrier. Such treatment often leads to significant improvements in mood and affect. Similarly, low folate levels strongly correlate with age-related cognitive decline and brain atrophy.Activated folate supports the myelin sheath protecting nerve fibers, highlighting its neuroprotective role. This highlights its vast therapeutic potential for long-term brain health and cognitive longevity.
Cardiovascular Protection via Active Folate
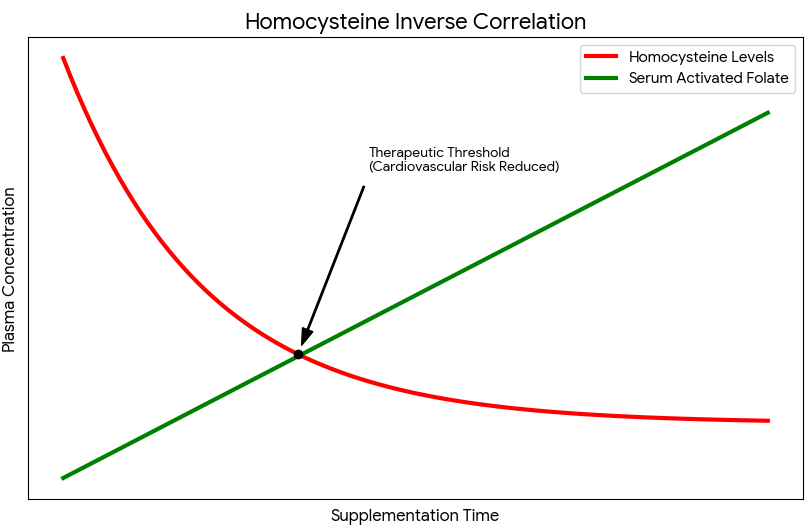
The cardiovascular benefits of activated folate center on its unique ability to regulate homocysteine, an amino acid that becomes toxic at high levels. Elevated levels of homocysteine in the blood act like a chemical abrasive, damaging the delicate endothelial lining of arteries and promoting the buildup of atherosclerotic plaque. This process drives heart disease, stroke, and heart attack, independent of cholesterol levels. The methylation cycle serves as the body’s primary mechanism for clearing homocysteine, converting it back into the essential amino acid methionine, which the body then uses for tissue repair. This conversion depends entirely on a methyl group donated by activated folate. Without this donation, homocysteine pools in the blood, creating a pro-inflammatory environment that dramatically degrades vascular health over time.
In individuals with MTHFR mutations, this clearance process basically stops. This leads to chronically high homocysteine levels that diet alone often cannot correct. Supplementing with activated folate directly fuels this conversion, effectively lowering homocysteine levels and mitigating cardiovascular risk. Clinical studies have demonstrated that 5-MTHF is significantly more effective than folic acid at lowering homocysteine in people with the C677T mutation. This offers a powerful, non-pharmaceutical intervention for preventing cardiovascular events. Furthermore, by improving endothelial function and reducing oxidative stress within the blood vessels, activated folate supports healthy blood pressure and overall circulatory resilience. The graph below visualizes the inverse relationship between 5-MTHF levels and plasma homocysteine, underscoring the direct impact this nutrient has on maintaining a healthy cardiovascular system.
 Prenatal Care and the Necessity of Activated Folate
Prenatal Care and the Necessity of Activated Folate
Perhaps the most well-known role for Vitamin B9 is in pregnancy, specifically for the prevention of neural tube defects like spina bifida. For decades, folic acid has been the standard of care recommended by governments and health organizations worldwide. However, the medical community is increasingly recognizing the superiority of activated folate for prenatal health, particularly given the prevalence of MTHFR mutations. By providing 5-MTHF, practitioners ensure that both mother and the developing fetus receive the bio-available methyl donors essential for healthy gene expression and cell division. This bypasses the risk associated with unmetabolized folic acid, which may block folate receptors in the placenta. The direct supply of activated folate ensures that the rapid cell division occurring in the first trimester proceeds without genetic errors or structural defects.
This direct supply of activat folate not only reduces the risk of neural tube defects.
Research has also linked it to a lower risk of other pregnancy complications, such as preeclampsia and recurrent miscarriage. Studies indicate that the body requires adequate methylation to vascularize the placenta properly; without it, the body can compromise placental function, leading to poor fetal growth.Furthermore, because pregnancy places a massive demand on the mother’s folate reserves, women with MTHFR mutations are at a distinct disadvantage if they rely solely on folic acid. They may ingest the recommended amount but fail to convert enough to support two lives. Therefore, modern prenatal vitamins are increasingly replacing synthetic folic acid with activated folate, acknowledging that the benefits extend throughout gestation and offer a safer, more effective insurance policy for the health of the baby.
Fertility and the Impact of Active Folate
Activated folate plays a crucial, often overlooked role in both male and female fertility. For women, folate status ensures ovarian function, egg quality, and endometrial preparation for embryo implantation. Poor methylation causes high homocysteine, which triggers uterine micro-clotting and prevents successful implantation. Optimizing methylation with active B9 improves the female reproductive environment. In men, spermatogenesis requires rapid cell division and immense DNA synthesis. Men with MTHFR mutations often exhibit lower sperm counts and higher DNA fragmentation, causing male factor infertility. Supplementing with activated folate significantly improves sperm parameters. This highlights the nutrient’s importance for both partners during preconception.
The connection between methylation and fertility underscores the necessity of addressing nutrient forms before conception even occurs. Many couples struggling with “unexplained infertility” may actually be dealing with undiagnosed MTHFR mutations and subsequent folate deficiencies. In these cases, synthetic folic acid may be insufficient to correct the imbalance. Switching to a protocol that includes high-quality activated folate can resolve these metabolic roadblocks, lowering oxidative stress in the reproductive system and enhancing the viability of gametes. It represents a simple yet powerful intervention that addresses the root biochemical causes of reproductive struggles. As fertility science advances, the focus is shifting toward optimizing the parental biochemical terrain, with active folate serving as a cornerstone of pre-conception care protocols.
Detoxification Pathways and Active Folate
In a toxic world, the body’s ability to detoxify environmental pollutants remains critical for maintaining health. The liver utilizes methylation to process and eliminate heavy metals, endocrine-disrupting chemicals, and excess hormones like estrogen.This Phase II detoxification pathway requires active B9; without it, toxins accumulate in fatty tissues and the brain. Such accumulation contributes to chronic fatigue, autoimmune conditions, and hormonal imbalances. For example, proper estrogen metabolism prevents harmful metabolite buildup, which researchers link to breast and ovarian cancers. Activated folate provides methyl groups to convert dangerous estrogens into safe, water-soluble compounds for excretion..
Furthermore, the production of glutathione, the body’s “master antioxidant,” is intrinsically linked to the methylation cycle. While glutathione production is technically part of the transsulfuration pathway, this pathway is downstream from the methylation cycle. If methylation stlls due to a lack of active folate, the production of glutathione often decreases as a compensatory mechanism. This leaves the cells vulnerable to oxidative damage and impairs the immune system’s ability to fight off infections. By restoring the methylation cycle with 5-MTHF, one indirectly supports glutathione synthesis, thereby enhancing the body’s overall detoxification capacity. This highlights why active folate is not just a vitamin for DNA, but a critical component of the body’s waste management and defense systems.
A Practical Framework for Activated Folate Supplementation
Understanding activated folate’s biochemical rationale is the first step; implementing it safely is the next. Market saturation with conflicting labels can confuse, but key principles can guide individuals toward optimal supplementation. B9 sources immensely influence biological outcomes. While leafy greens provide natural folates, those with genetic variants struggle to achieve therapeutic levels through diet alone.Optimizing health requires avoiding synthetic folic acid, given its conversion issues and the prevalence of MTHFR mutations. High-quality supplements providing activated folate—often labeled L-Methylfolate, L-5-MTHF, or 6(S)-5-MTHF—represent the most effective choice.
When selecting a supplement, quality and the specific isomeric form are non-negotiable factors. The “L” or “6(S)” prefix is critical, as this indicates the biologically active isomer that fits into the body’s receptors.The biologically inert “D” form mirrors the active molecule and may compete for absorption, reducing efficacy. Reputable supplements often use patented Quatrefolic® or Metafolin®, clinically studied for stability, solubility, and bioavailability. These patented forms deliver pure activated folate that resists stomach degradation and enters the bloodstream intact. Consumers must read labels carefully, as many formulas still “fairy dust” with cheap folic acid, undermining active ingredients.
Dosing Strategies for Active Folate
Transitioning to this potent nutrient requires a personalized approach, as individual responses vary significantly. Efficient activated folate can rapidly kick-start dormant methylation pathways, causing over-methylation in sensitive individuals. Sudden surges in neurotransmitter production and detoxification cause anxiety, irritability, headaches, or insomnia.The guiding principle “start low and go slow” ensures safe supplementation. A low starting dose, like 400 mcg, allows the body to adapt without overwhelming the system. Users should monitor dosing and adjust gradually under a healthcare professional who understands methylation genetics.
Individuals with significant MTHFR blockages or high homocysteine may eventually require higher doses. Clinicians must titrate these doses, sometimes reaching 1mg to 15mg daily, with extreme care. Lifestyle factors like stress and diet deplete activated folate, changing dosing requirements over time. Monitoring symptoms remains key. If agitation occurs, niacin can “mop up” excess methyl groups, acting as a metabolic brake. This dosing nuance highlights that activated folate acts as a powerful metabolic intervention, not a passive ingredient. Understanding this potency allows users to harness benefits while minimizing side effects through a tailored wellness approach.
The Synergistic Role of B12 and Activated Folate
Finally, one must never consider activate folate in isolation, as its function has an inextricable link with Vitamin B12. Folate and B12 are synergistic partners in the methylation cycle, working in tandem to convert homocysteine to methionine. The enzyme methionine synthase requires B12 (in the form of methylcobalamin) to accept the methyl group from folate. Taking high doses of activated folate without ensuring adequate B12 status can lead to a condition known as the “folate trap.” In this scenario, folate becomes metabolically stuck inside cells, unable to complete its cycle because B12 is not present to accept the methyl group. This effectively renders the folate useless despite high blood levels, halting the methylation process.
Folate supplementation can mask B12 deficiency’s hematological signs while allowing neurological damage to progress unchecked. This can lead to serious, sometimes irreversible, nerve damage. Therefore, any activated folate protocol must include active B12, like methylcobalamin, to maintain methylation cycle synergy. One should avoid cyanocobalamin for the same reasons as folic acid; its conversion steps can be inefficient. Combining active folate and B12 ensures smooth methionine synthase operation, protecting both the blood and the brain.
Conclusion: The Biological Necessity of Activated Folate
Rigorous biochemical science provides overwhelming evidence for shifting toward activated folate. This shift moves away from imprecise mass folic acid fortification toward personalized, genetically informed medicine. Understanding individual biochemistry and testing for MTHFR variants helps us prioritize activated folate and bypass metabolic hurdles. Using the body’s finished product supports brain health, protects the cardiovascular system, and unlocks cellular wellness. Clinical outcomes support activated folate’s superiority as a matter of biological fact. This key fits the intricate lock of human methylation, making it the definitive choice for modern vitality.
A consistent supply of this nutrient is a cornerstone of this new approach to preventative health. Expectant mothers, patients with depression, and those reducing heart disease risk must respect the body’s enzymatic limitations. Clinical recommendations are clear: the body needs activated folate for cellular delivery, detoxification, and optimal gene expression. Embracing this bio-identical nutrient aligns our supplementation strategies with our biology. This ensures we provide our cells with the precise tools they need to flourish.

Comments are closed.